26 Draw The Orbital Diagram For The Ion Co2+. Wiring Database 2020
The Octet Rule The other halogen molecules (F 2, Br 2, I 2, and At 2) form bonds like those in the chlorine molecule: one single bond between atoms and three lone pairs of electrons per atom. This allows each halogen atom to have a noble gas electron configuration.
40 co3+ orbital diagram
The n + l n + l rule tells you the order in which atomic orbitals are filled, and according to the rule the 4s 4 s orbital is occupied before the 3d 3 d orbital because it has lower energy. Thus, the electron configuration of Mn M n is [Ar]3d54s2 [ A r] 3 d 5 4 s 2 while that of Co C o is [Ar]3d74s2 [ A r] 3 d 7 4 s 2 .

Electron Configuration for Co, Co2+, and Co3+ (Cobalt and Cobalt Ions
The electron configuration of an atom of any element is the of electrons per sublevel of the energy levels of an atom in its ground state . This handy chart compiles the electron configurations of the elements up through number 104.

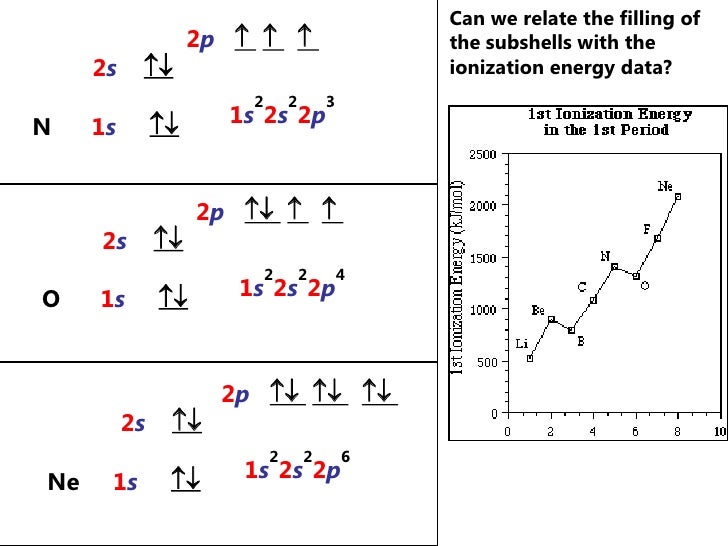
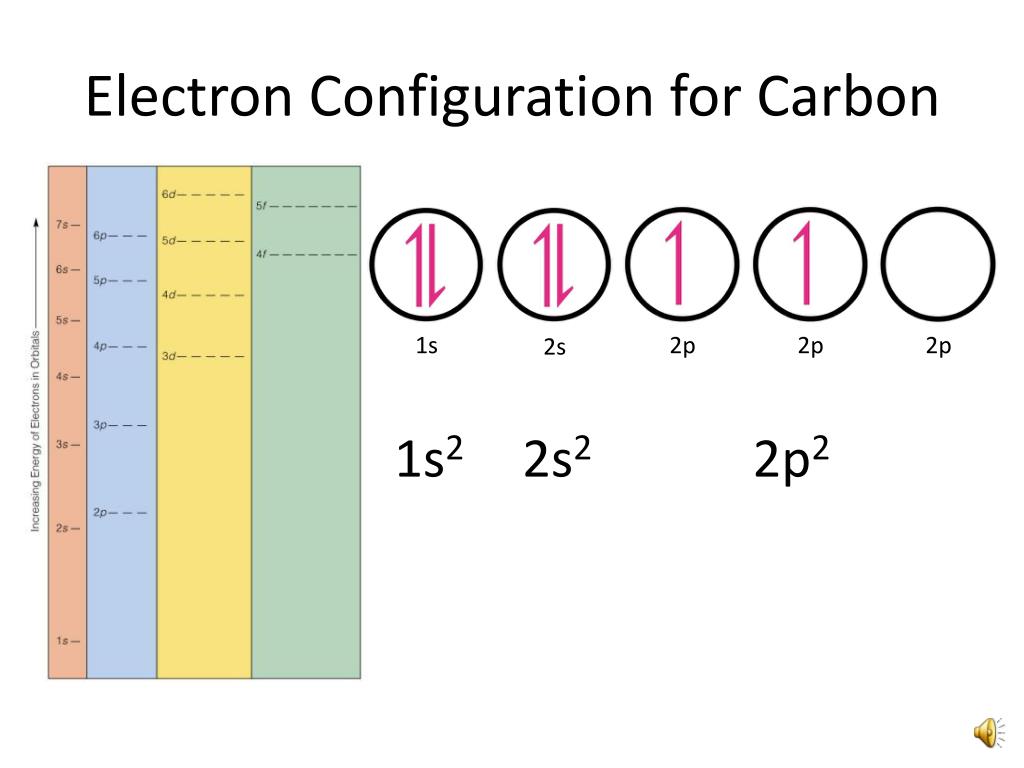
Carbon Electron Configuration
In this case, 2+2+6+2+6+2+10+6+2+1= 39 and Z=39, so the answer is correct. A slightly more complicated example is the electron configuration of bismuth (symbolized Bi, with Z = 83). The periodic table gives the following electron configuration: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p65s2 4d10 5p6 6s2 4f14 5d10 6p3.

Electron Geometry for CO2 (Carbon Dioxide) YouTube
An electrochemical conversion of carbon dioxide into chemical fuels is a promising approach to store the renewable energy sources 2. However, a critical challenge toward efficient CO 2 reduction.
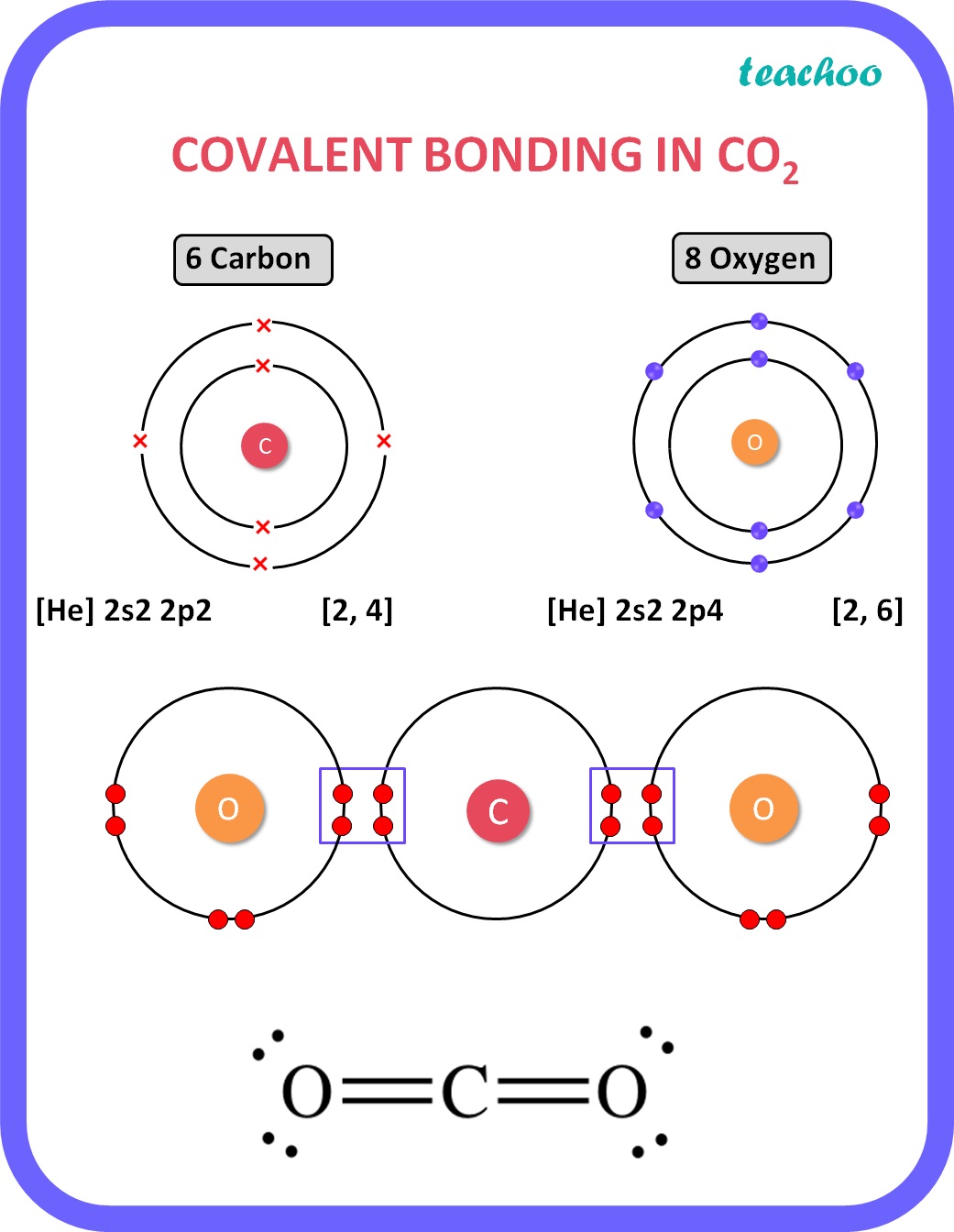
[Class 10] Electron dot structure of carbon dioxide with formula CO2
The electron configuration of Cobalt is [Ar]4s 2 3d 7. When observing Cobalt 3+, we know that Cobalt must lose three electrons. The first two to go are from the 4s orbital and Cobalt becomes:[Ar]4s 0 3d 7. Then, the next electron leaves the 3d orbital and the configuration becomes: [Ar]4s 0 3d 6. Thus, we can see that there are six electrons.
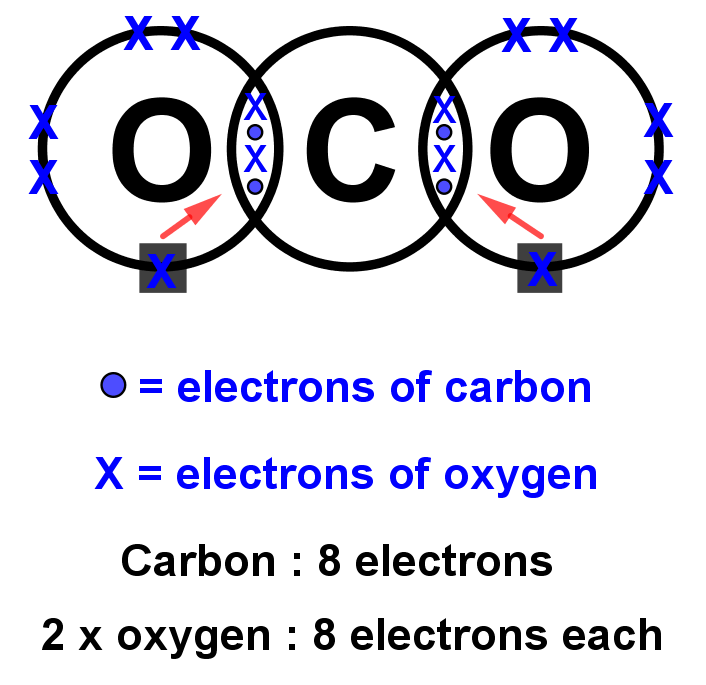
13+ Co2 Dot And Cross Diagram Robhosking Diagram
Hello Guys,Determining the electron configuration of any element is an easy and quick process, given that you know all the required information and general c.
PPT Orbital Filling Electron Configurations PowerPoint Presentation
What is the electron configuration of cobalt? The total number of electrons in cobalt is twenty-seven. These electrons are arranged according to specific rules in different orbitals. The arrangement of electrons in cobalt in specific rules in different orbits and orbitals is called the electron configuration of cobalt.

Orbital Diagram For Cobalt
To write the configuration for the Cobalt ions, first we need to write the electron configuration for just Cobalt (Co). We first need to find the number of.

Electronic Configuration for Carbon spdf Trick Chemistry Atomic
The electron configuration of Co is 1s2 2s2 2p6 3s2 3p6 3d7. Co requires three more valence electrons in its d-orbital to attain full filled configuration. What is the complete electron configuration of Co 2+? The complete electron configuration of Co2+ is 1s2 2s 2 2p6 3s2 3p6 3d7.Co2+ have 7 valence electrons in the subshell.
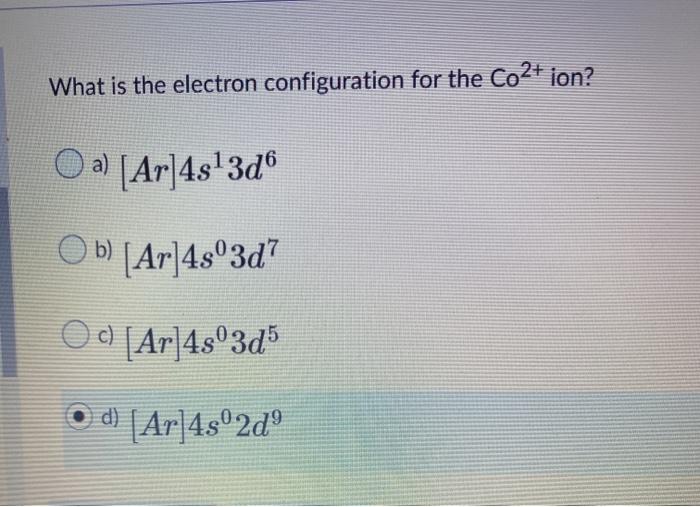
Solved What is the electron configuration for the Co2+ ion?
The electron configuration and the orbital diagram are: Following hydrogen is the noble gas helium, which has an atomic number of 2. The helium atom contains two protons and two electrons. The first electron has the same four quantum numbers as the hydrogen atom electron ( n = 1, l = 0, ml = 0, ms = +12 m s = + 1 2 ).

Carbon Electron Shell Diagram
To determine the number of valence electrons for CO2, the Carbon dioxide molecule, we'll use the Periodic Table. Organizing the Periodic Table by Group, ski.
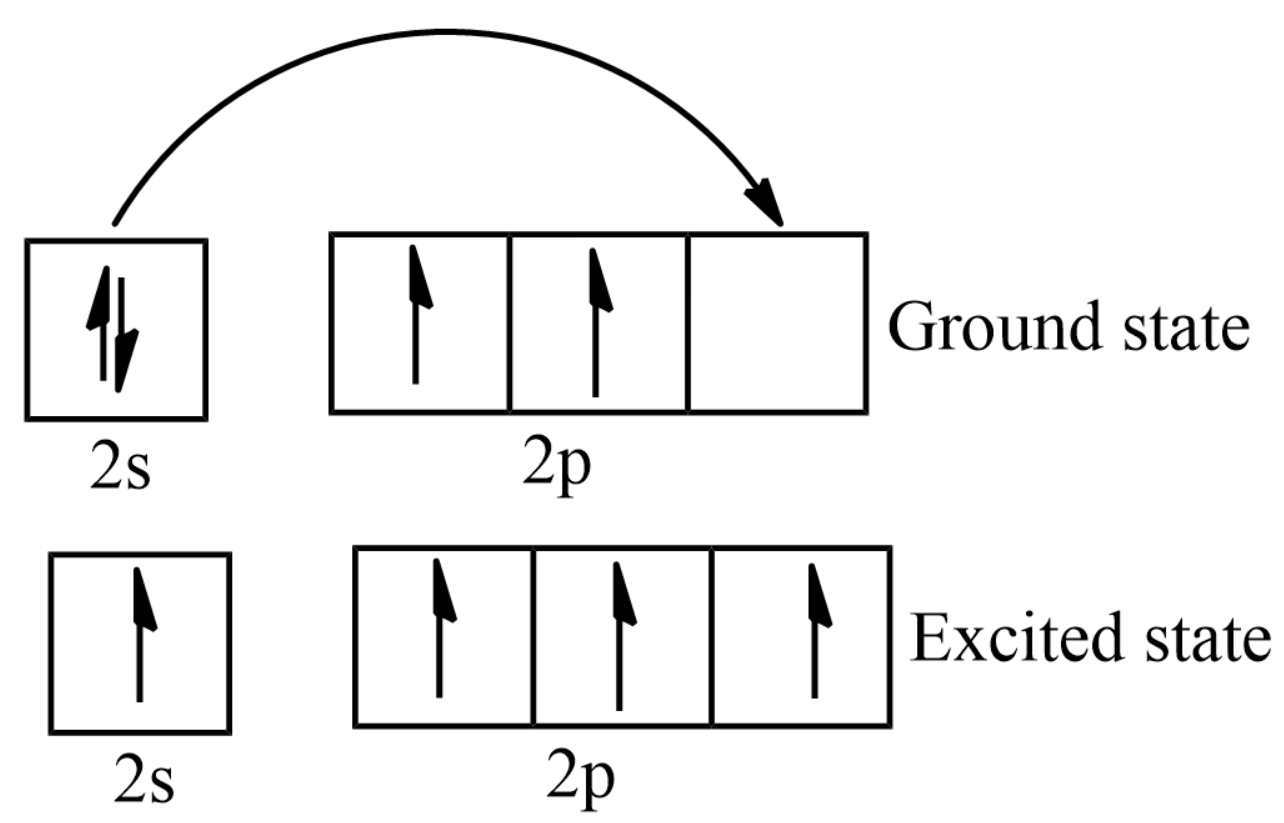
What is the excited state of carbon?
With ever-increasing atmospheric carbon dioxide concentrations and commitments to limit global temperatures to less than 1.5 °C above pre-industrial levels, the need for versatile, low-cost.
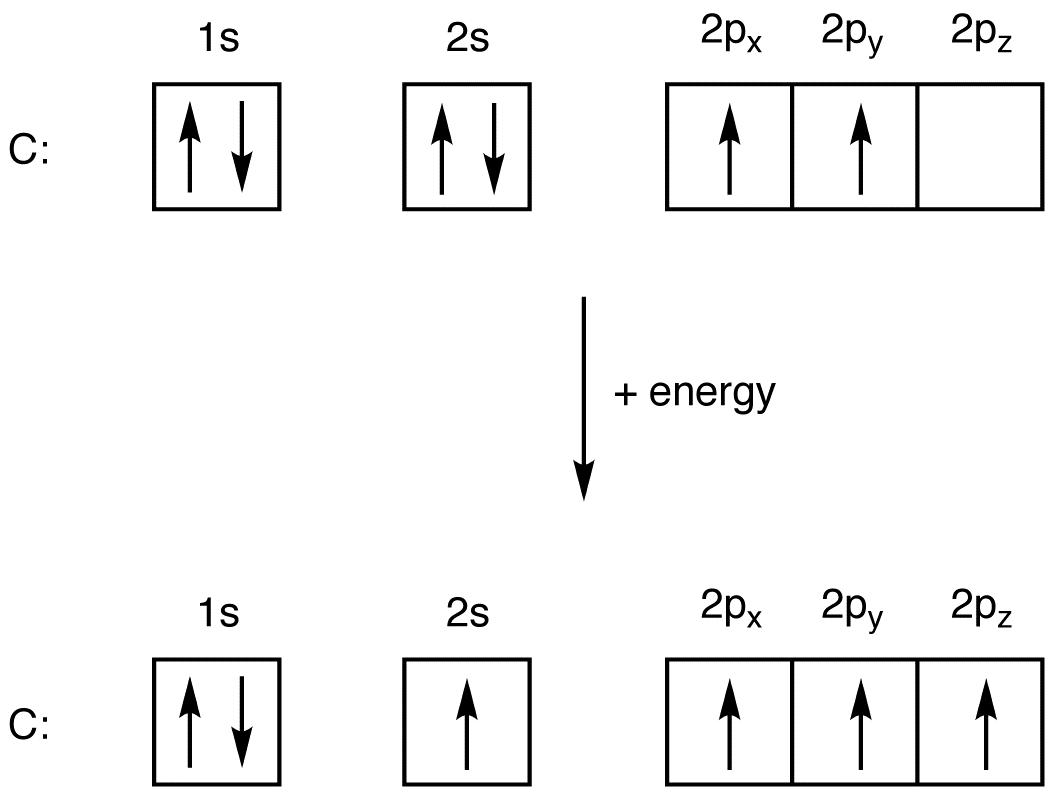
Electron Configuration Of Carbon
Losing the two 4s electrons leaves a positive charge of +2 and seven 3d electrons. Answer link. Co^ (2+) would most likely be 1s^2 2s^2 2p^6 3s^2 3p^6 4s^0 3d^7, and Co would have 4s^2 instead (the rest otherwise the same). The two outermost electrons are the 4s electrons so these are the two electrons that most likely will be lost.
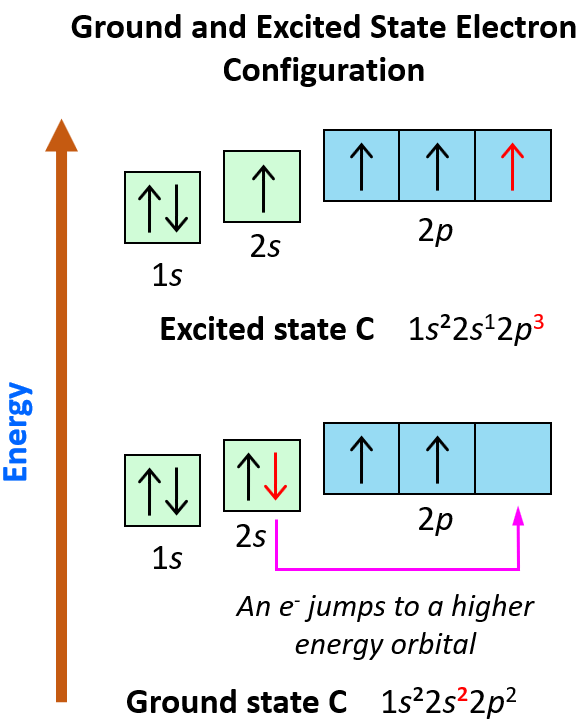
Electron Configurations Chemistry Steps
Here, the electron configuration of cobalt ion(Co 2+) is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 7. This cobalt ion(Co 2+) has twenty-seven protons, thirty-two neutrons, and twenty-five electrons. Also, cobalt has one more ion. That is Co 3+. Co - 3e - → Co 3+ Here, the electron configuration of cobalt ion(Co 3+) is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 6.
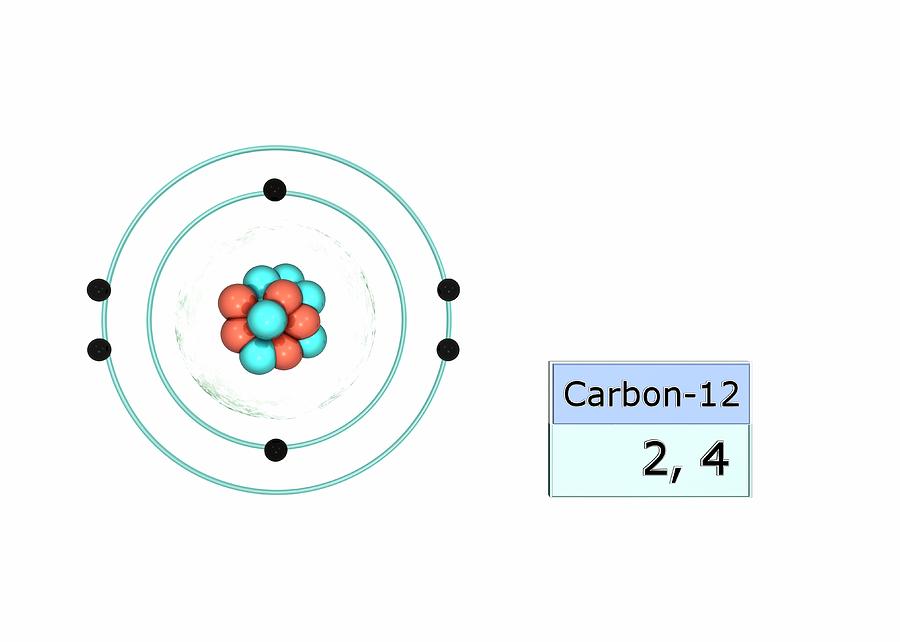
Carbon Electron Configuration Photograph by Photo
Answer: The electron configurations of the elements are presented in Figure 2.2.3, which lists the orbitals in the order in which they are filled. In several cases, the ground state electron configurations are different from those predicted by Figure 2.2.1. Some of these anomalies occur as the 3 d orbitals are filled.